Research Projects in the Center for Informatics and Telehealth in Medicine (InTelMed): Overview
- PHM for Orbiting and Surface-based Space Habitats (NASA Gateway/Artemis)
- PHM for Human Health & Performance
- PHM for Astronauts
- Bidirectional data transfer framework between sensors/smartphones and the cloud
- Big data mining tools in the medical field at large, including data analytics, diagnostics, and prognostics
- Image (both still and video) processing and analysis
- Disruptive electric neural stimulation for artificial vision implants (epi-retinal, sub-retinal, supra-choroidal)
- Mobile smartphone-based ophthalmic examination devices (EFFORT OUTSOURCED TO STARTUP COMPANY)
- Optical extensions for mobile ophthalmic examination devices (EFFORT OUTSOURCED TO STARTUP COMPANY)
PHM for Orbiting and Surface-based Space Habitats (NASA Gateway/Artemis)
With NASA’s push for prolonged stays aboard orbiting or surface-based space habitats (e.g., NASA Gateway/Artemis programs) on the Moon and soon on Mars, there is a need to monitor these space habitats for their viability, likely effected through a heavy reliance on abiotic sensors embedded in a Prognostics and Health Management (PHM) framework to detect anomalies aboard these space habitats. Recently, we introduced the concept of employing the crew member as a biosensor to be integrated into the overarching space habitat PHM system to increase the breadth of available data when monitoring environment health.
PHM for Human Health & Performance
Prognostics and Health Management (PHM) is traditionally applied in aerospace to help extend the remaining useful life of high value aerospace assets (i.e., airplanes, jet engines, etc.) at significantly reduced cost of maintenance. By analogy, astronauts, pilots, warfighters, and athletes, etc. are considered high value human assets because of the enormous financial investment in their education and (continued) training. As such, over the past several years, PHM has been increasingly infused by our group into the human healthcare, precision medicine, and human performance sectors. With the overwhelming emergence of wearable smart sensors, sensor-data-fusion, big data mining, machine/deep learning, diagnosis and prediction, and electronic health records and databases, PHM has found an entirely new application field: PHM for human health & performance on Earth and in Space.
PHM for Astronauts
Crew health and performance are critical to successful human space exploration. Long-duration space missions bring numerous risks that must be understood and mitigated in order to keep astronauts healthy, rather than treat a diagnosed health disorder. Crewed missions venturing beyond Low Earth Orbit (LEO) and beyond (e.g., travel to the Moon and Mars) will require technology solutions for crew health care to address physiological, psychological, performance, and other needs in-situ, e.g., self-sufficiency, as an emergency or quick-return option will not be feasible. Therefore, onboard capabilities that would allow for early self-diagnosis of impending health issues, and autonomous identification of proper responses on negative trends to keep astronauts healthy are critical. With the absence of real-time medical ground support, personal health-tracking tools for health monitoring, health risk assessment and management are required for any crew member to predict her/his future health condition if no preventive measures are taken. It is important to stress that the framework of conventional medicine and even the framework of telemedicine are not viable/feasible as a healthcare concept for crew members especially for long-duration space missions (e.g., flight to Mars) and eventual human settlement on other planetary bodies (e.g., Moon and Mars) where support and intervention from ground control on Earth is prohibitive because of distance. As such an autonomous healthcare concept is essential based on Prognostics and Health Management (PHM) principles to ensure crew health and ultimately mission success. Rather than treating problems medically, it is prudent to prevent them from happening in first place and/or to catch them early on where mitigation and interventions are still straightforward and not involved.
Bidirectional data transfer framework between sensors/smartphones and the cloud
What is currently lacking in modern telemedicine is the capability of in-situ, near real-time analysis and diagnosis of (image) data obtained with sensor of smartphone-based examination devices. In order to establish a true smart service platform, a server-based telediagnostic analysis capability for current and future smartphone-based examination devices is critical. Such a server-based telediagnostic analysis capability requires a bidirectional data transfer framework/protocol that is HIPAA-compliant (through encryption), reliable, and fault-tolerant. As part of the NSF Partnership for Innovation (PFI): Building Innovation Capacity (BIC) grant (2014–2018) titled “Making Full Use of the High-Resolution Image Capability of Smartphones to Collect Data through Ophthalmic Devices for Smart Mobile- and Tele-Health”, Dr. Fink’s lab has created such as bidirectional data transfer framework/protocol.
Big data mining tools in the medical field at large, including data analytics, diagnostics, and prognostics
A suite of classification algorithms (i.e., Automated Global Feature Analyzer (AGFA)) has been developed that (1) solves the sensor data fusion problem for biomedical data by extracting objective parameters from the biomedical data and fusing them into a mathematical feature vector; (2) allows for the comparison of biomedical data sets via their respective feature vectors, e.g., both intra- and inter-patient; (3) captures the time-development of a disease due to disease progression or treatment; and (4) provides the foundation for subsequent disease detection and identification through machine learning and deep learning algorithms.
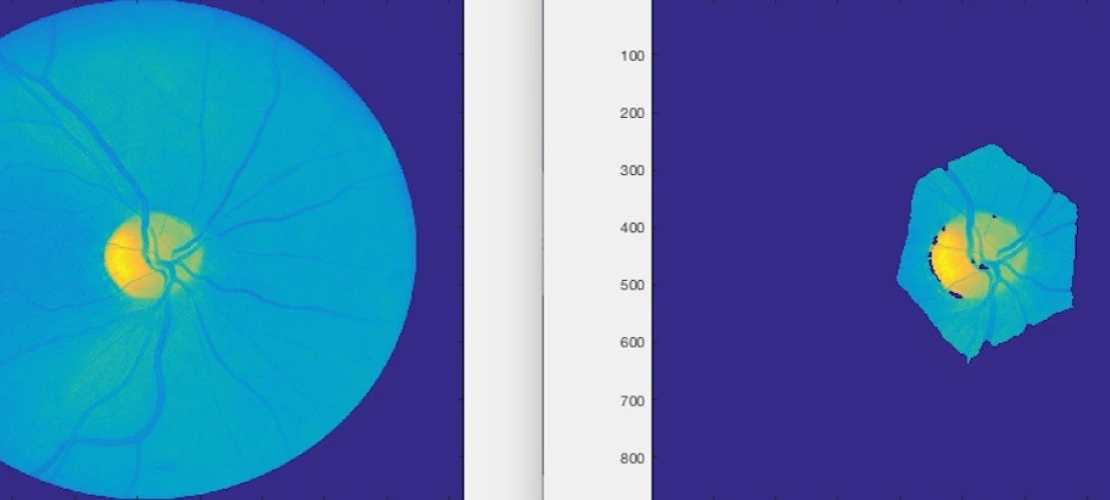
Image (both still and video) processing and analysis
Dr. Fink’s lab has been developing the Automated Global Feature Analyzer (AGFA), which performs target identification using image segmentation, feature extraction, classification, unbiased anomaly detection (i.e., based on feature space only), and target prioritization within mapped or imaged areas or regions. AGFA enables automated science analysis and anomaly detection within imaging data.
Disruptive electric neural stimulation for artificial vision implants (epi-retinal, sub-retinal, supra-choroidal)
Currently, there is a tremendous interest in treating a subject having a clinical condition associated with impairment in electrical stimulation of cells or tissues, thereby “a clinical condition associated with impairment in electrical stimulation” refers to a manifestation of a clinical condition due to improperly functioning neurons or retina. Such improper function can be due to impaired transmission of a signal from one cell to another cell (e.g., typically to an adjacent cell), lack of cell function even in the presence of a properly transmitted signal from another cell, or due to cell death or disrupted pathways.
Many prostheses have been developed to treat spinal cord injury by implanting an artificial electric stimulation device. Exemplary clinical conditions associated with impairment in electrical stimulation of cells and/or tissues include, but are not limited to, neural cell damage or impaired neural cell function such as retinal damage (such as retinitis pigmentosa, retinal detachment, diabetic retinopathy, and macular degeneration), optic neuropathy, glaucoma, stroke, spinal cord injury, peripheral nerve injury, demyelinating disease (such as multiple sclerosis), and central nervous system injury secondary to ischemia, compression, nerve injury, infection affecting nerve cell function, elevated intracranial pressure, elevated intraocular pressure (ocular hypertension), congenital and hereditary genetic diseases associated with impaired nerve cell function, toxic neuropathy and encephalopathy, neurological sequelae of systemic diseases such as chronic arterial hypertension, diabetes, HIV infection, systemic lupus, coagulation disorders, Parkinson's disease, Alzheimer's disease, prion disease, and paralysis. Retinal damage or impaired retinal function can lead to diminished sight and blindness. And, as the age of the general population increases, the number of people suffering from diminished sight due to these causes increases.
Functions of some cells can be replaced or approximated by providing an appropriate electrical stimulation to cells they are connected to. It should be appreciated that as used herein, a clinical condition associated with electrical stimulation impairment refers to any clinical condition that manifests itself due to an improper or missing signaling of one cell to another cell, often an adjacent cell. Several devices have been developed to attempt to restore vision loss due to retinal damage. For example, photovoltaic devices, which are attached to a portion of a retina, have been developed to replace the signals that normally emerge from rods and/or cones in a healthy eye within the retina by stimulating functioning cells. Although such devices may provide some stimulation, the devices suffer from several drawbacks.
Vision impairment can be caused by numerous factors. While many vision impairments can be corrected by corrective eyewear and surgery, not all vision impairment can be treated by such relatively simple methods. For example, some vision impairments involve problems with the light-processing functions of the eye. These problems are usually caused by abnormalities of the retina and macula such as retinitis pigmentosa and age-related macular degeneration. Vision impairments due to these causes cannot be addressed with corrective eyewear or eye surgery. It is estimated that globally over 1.5 million people have progressive vision loss as a result of retinitis pigmentosa, the primary cause of inherited blindness.
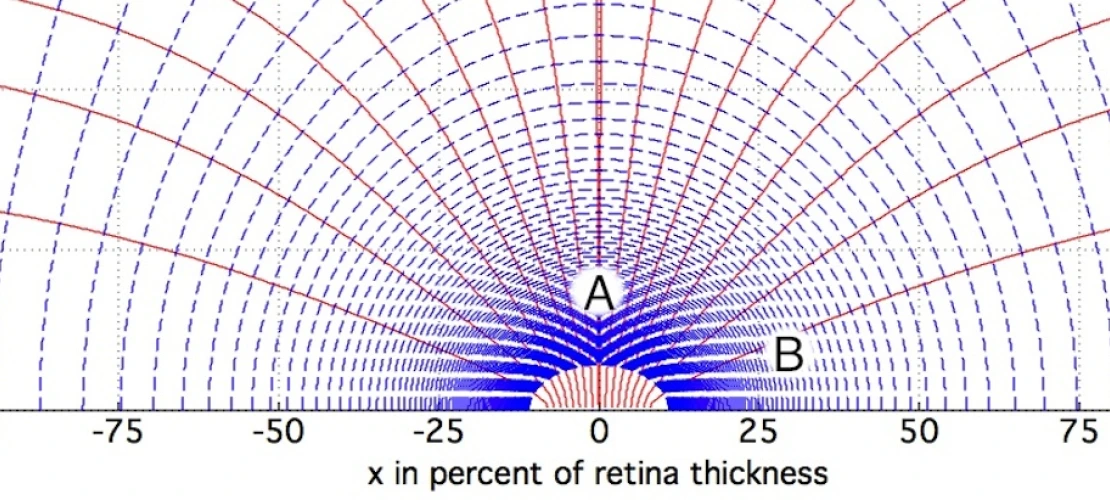
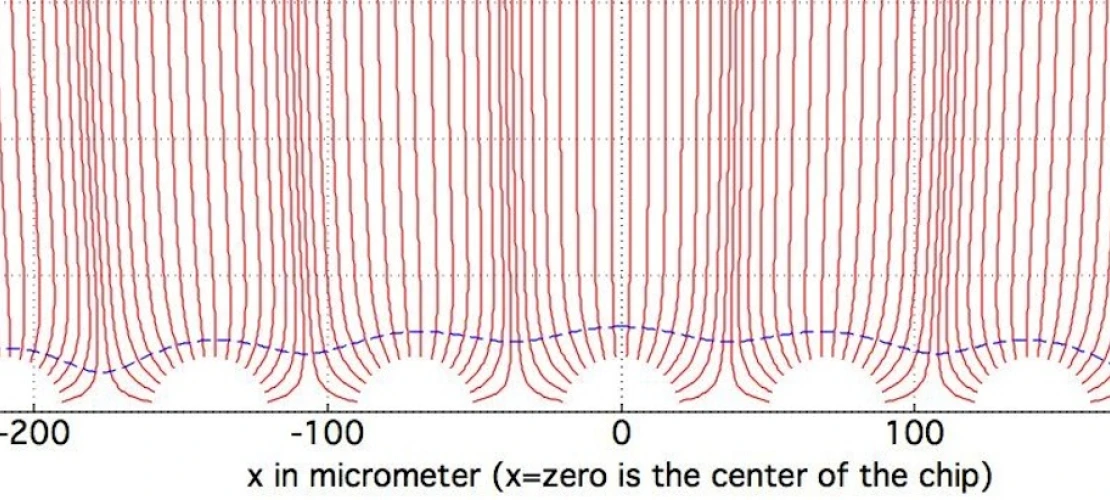
To address such vision impairment, research on retinal implants has been ongoing for about two decades. One area of such research is to restore a small part of vision to people suffering from blindness due to retinitis pigmentosa or due to age related macula degeneration. One of the conventionally available devices for treating vision impairment is a chip with an array of electrodes that is placed into an epi-retinal, sub-retinal, or supra-choroidal position. With this device, electric currents emerging from the electrodes are seen by the blind person as small phosphenes. Thus, electrodes serve as pixels for presenting an image. Unfortunately, despite some encouraging results the goal of presenting a gray-scaled picture with a thousand or more pixels has not yet been reached. Other devices that have been developed include optic nerve implants, lateral geniculate nucleus implants, cortical implants, as well as non-invasive remedies such as electric tongue stimulators, and tactile stimulators.
Problems associated with currently available retinal implants include use of too simple time profiles of the electric stimulation signals. Currently, the most common time profile is the monophasic rectangular voltage pulse, which yields a biphasic current pulse. Or the biphasic rectangular voltage pulse, which yields a triphasic current pulse. Such pulses lead to the depolarization (or hyperpolarization) of a cell membrane in the first phase of the current, and to a polarization of opposite sign in the next phase of the current.
Another shortcoming in many conventional retinal implants is using only one electrode per pixel. Moreover, many research groups are using a common remote counter electrode that is far away from the electrode array. While one group in Australia is using six (6) counter electrodes around a center electrode on a hexagonal grid and, by current splitting, this group also uses a common remote counter electrode, i.e., far away from the electrode array. One of the key problems associated with using counter electrodes at infinity, is that it leads to cross-talk.
Yet another problem associated with conventional retinal implants involves too little effort for shaping the electric field (or current). Simultaneous firing of neighboring electrodes leads to bunching of field lines, i.e., increased density of field lines above the electrodes. But it also means undesired cross-talk between neighboring electrodes.
Therefore, there is a need for a novel neural electrical stimulation paradigm and its optimization, which will be essential for achieving visual perception at a spatial resolution comparable to the number of electrodes on the implanted electrode arrays (i.e., generation of discrete phosphenes) – especially when increasing the electrode density of the implanted electrode arrays of epi-retinal, sub-retinal, and supra-choroidal vision implants. This novel stimulation paradigm uses ultra-short (i.e., microseconds) electric stimulation, electric field shaping, and shifting multi-poles, and is potentially applicable to neural stimulation beyond vision, such as paralysis and deep brain stimulation.
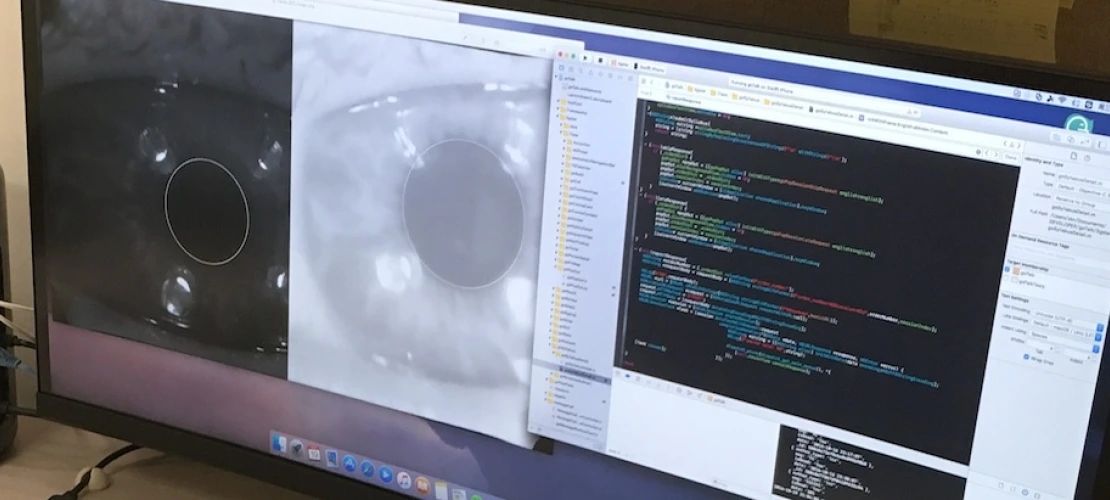
Mobile smartphone-based ophthalmic examination devices (EFFORT OUTSOURCED TO STARTUP COMPANY)
Mobile Health and Tele-Health are strongly growing markets worldwide (~$38 billion by 2022) with a significant savings potential in healthcare costs to patients, health insurers, and economies at large. Dr. Fink was awarded a $0.8 million NSF Partnership for Innovation (PFI): Building Innovation Capacity (BIC) grant (2014–2018) titled “Making Full Use of the High-Resolution Image Capability of Smartphones to Collect Data through Ophthalmic Devices for Smart Mobile- and Tele-Health.” The focus of this effort was on tele-ophthalmology, i.e., to bring the ophthalmic healthcare examination to the patient rather than the patient to the clinic, thus enabling early detection of diseases and timely intervention to prevent irreversible vision impairment. This tele-ophthalmic paradigm serves especially those who are geographically dispersed or operate/live in austere environments, where time, cost, and possibility of travel make access to even adequate medical care difficult if not impossible.
Optical extensions for mobile ophthalmic examination devices (EFFORT OUTSOURCED TO STARTUP COMPANY)
As part of the NSF Partnership for Innovation (PFI): Building Innovation Capacity (BIC) grant (2014–2018) titled “Making Full Use of the High-Resolution Image Capability of Smartphones to Collect Data through Ophthalmic Devices for Smart Mobile- and Tele-Health”, Dr. Fink’s lab has turned smartphones into smart mobile healthcare data acquisition devices, equipped with specialized optics and tele-medical analysis and diagnosis capabilities via a smart server-backend, for mobile- and tele-health applications.